Applications
We cover a large variety of appplications

Freeze Dryers
Autoclaves
freezers
refrigerators
ovens
Temperature Mapping Equipment
Pharmaceutical, Biotechnology, Food Industry Aplications
Full list of applications

Autoclaves
Autoclaves are essential for the Life Sciences Industry. The validation of sterilization cycles is critical to ensure sterility of product, components and instruments, amont others.

Cold chain shipments
Tracking the temperature of a product during cold chain shipping is a problem for many in the pharmaceutical industry. If insulated containers fail to properly keep a product at its designated temperature, the product may be ruined by the time it arrives. When this happens, it is costly and a waste of time.

Cold rooms
Cold room mapping is a simple application that can be handled with various technologies. The XpertVal® thermocouple-based system is a cost effective solution for application having many test points in close proximity.

Lyophilizers (Freeze-dryers)
The extreme low temperatures and limited shelf clearance in lyophilizers make thermal mapping with conventional equipment rather difficult. That is why we developed the GMPF LP RF low-profile temperature loggers.

Freezers
Freezer temperatures may vary and typically range from -25°C to -10°C. Regulations require that these systems be periodically mapped to ensure that they continue to fulfill their intended purpose.

Incubators
Incubator mapping can be easily performed with a combination of temperature and CO2 data loggers. The GMPF CO2 data logger records and transmits temperature and CO2 data.

Logistics
Logicstics require various types of validations and mappings, starting with standard temperature monitoring, to pressure and temperature or humidity and temperature recording.

Microbial Detection Systems

Ovens
Ovens are widely used for drying, warming, sterilization, melting, baking various sub-products, parts, substances or final products and they must maintain the specified temperature.

Package integrity
With Lives International, you can monitor the package integrity during all points of shipment.

Refrigerators
The reliability of refrigerators is crucial for pharmaceutical and biotech companies, as temperature-sensitivite products are stored in cold environments. Refrigerators used for these purposes must be periodically validated to ensure they are keeping products at the proper temperature no matter where in the refrigerator they are placed.
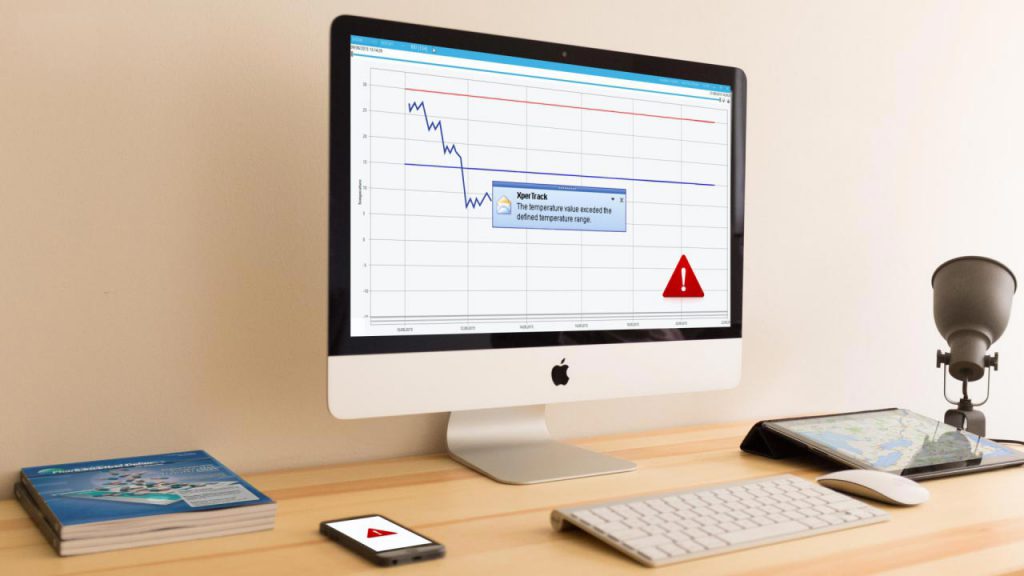
Remote monitoring
When you’re shipping your temperature-sensitivie product, you are aware of all the things that can go wrong. The product can be too warm or cold, be dropped and broken, or be lost altogether. The solution for all of these problems is remote monitoring for cold chain shipments, such as the XperTrack® system from Lives International.

Rubber stopper washing machine
Rubber stopper washing machines are widely used in pharma industry and they require validation of the washing/sterilization cycle. Most of the stopper washers available today combine multiple features, like washing, sterilizing, hot steam drying and siliconizing rubber stoppers, aluminium caps, plungers, all kinds of disk seals and closures.

Shipping Studies
Shipping studies are needed to evaluate whether the shipping process meets specified product storange conditions.
Our GDP temperature and humidity loggers are ideal for this task.

Stability chambers
ICH Q1A provides guidance for the stability testing of new drug substances and products.

Steam in place (SIP)
The span of large steam in place systems can make the installation of thermal validation thermocouples very difficult, if not impractical. Wireless data loggers can provide a practical alternative for this application due to the simplicity of their installation.

Sterilization tunnels
Sterilization tunnels(depyrogenation tunnels) are important in ensuring pharmaceutical equipment is sterile by moving equipment through a high-heat tunnel at a predetermined rate. These tunnels must be validated periodically to ensure that the head inside is at the rate it should be.

Ultra low freezers
Ultra low freezers or ULT freezers are typically used to store contents between -40 to -86°C. ULT freezers are used for long term storage of proteins, cell extracts or reagents, and thus they require periodic validation.
